CYTOTRON
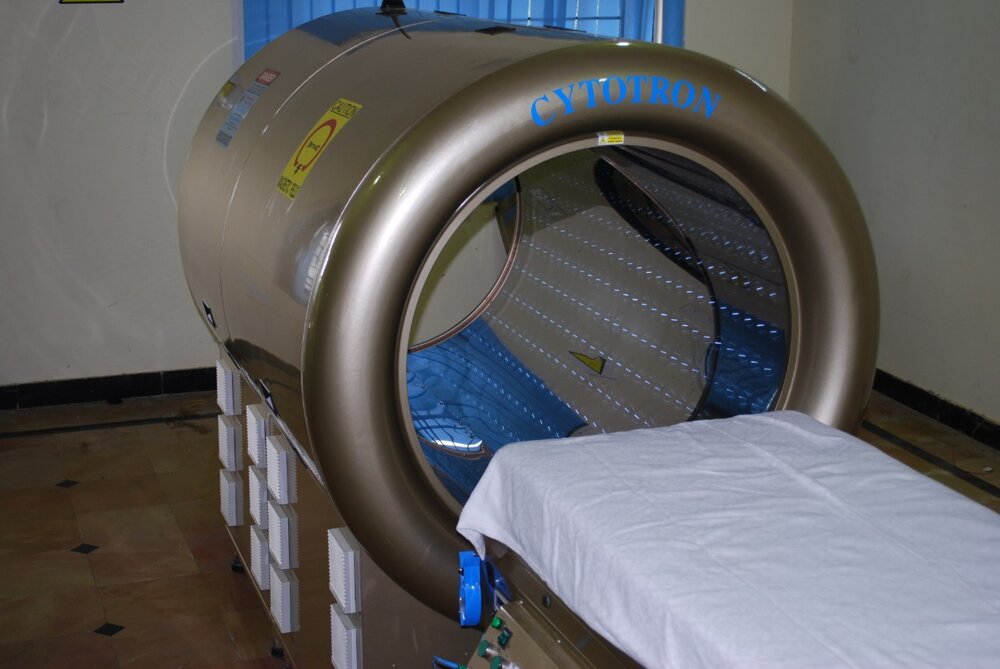
The Cytotron® is a non-invasive whole-body therapeutic device that is a breakthrough in tissue engineering.
The Cytotron® is a stand-alone device based on Rotational Field Quantum Magnetic Resonance (RFQMR) platform technology that enables whole body therapeutics called Quantum Magnetic Resonance Therapy (QMRT). QMRT is the Cytotron® device-mediated, an innovative therapeutic modality for tissue Degeneration (for Cancer) and Regeneration (for human degenerative disease indications), currently in the clinical stage of development in the US. The Cytotron® operates at the safe end of the Electromagnetic Spectrum, and therapeutic signals are lower than microwave and cellphone frequencies.
RFQMR Technology in a nutshell
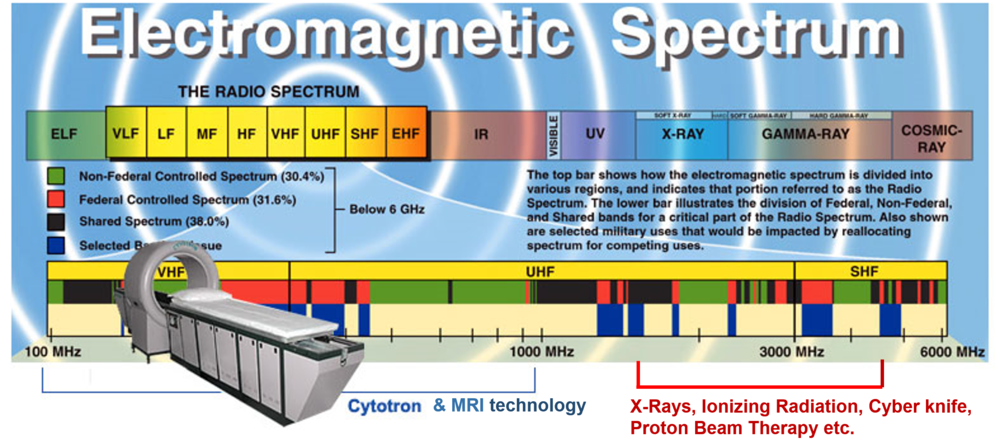
Cytotron Facts
Tissue Degeneration – Cancer
On October 24th 2019, the Cytotron was granted Breakthrough Device Designation by the U.S. FDA for Breast, Liver, and Pancreatic Cancers. (LINK) Additional solid tumor indications of use and other life-limiting indications are being pursued in parallel.
The Cytotron® device delivers rotating, target-specific, modulated, safe Radio Frequencies in the presence of a pulsed, integrated, instantaneous magnetic field. The presumed modulation of the transmembrane potential of tumor cells and downstream cellular signaling by RF for tissue degeneration in cancer underlies Rotational Field Quantum Magnetic Resonance platform technology. Whole-body MRI for tissue proton density determinations is used to compute individualized dosimetry to target solitary or multiple regions of interest in the whole body, simultaneously. Exposure to QMRT is for 1 hour daily for 28 consecutive days. Quality of Life assessments, overall survival, and tumor stability endpoints have shown remarkable improvement over standard of care related treatment outcomes (see publication in a peer-reviewed journal in Resources).

Cytotron for the Pediatric Brain Tumor Clinical Trial at National Children’s Hospital (HIM) in Mexico City
